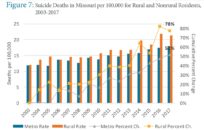
JEFFERSON CITY, Mo. — Missouri is one of the first states to submit a COVID-19 vaccine distribution strategy to the Centers for Disease Control and Prevention (CDC), according to Gov. Mike Parson.
“This has been an incredible collaborative effort, and I want to thank all of the agencies and partners involved for their hard work and dedication,” Parson told reporters Thursday. “In the midst of an ever-changing and unprecedented situation, our team of professionals has done outstanding work to develop a detailed plan. We are in a great place in the planning process and will be well-prepared to take action as soon as a vaccine becomes available.”
The plan was based on guidelines published last month by the CDC.
Department of Health and Senior Services Director Dr. Randall Williams said the CDC is expected to submit its feedback on the plan by Oct. 26.
Missouri will distribute the first round of vaccines to health care facilities, starting with long-term care facility staff and other health care workers. As availability increases, the state intends to spread its focus to the elderly and those at higher risk of serious complications due to preexisting health conditions.
The vaccine will be distributed to schools and “critical businesses” at the same time, with the final phase of distribution opening to the general public.
The state will partner with local governments, community groups, hospitals, and the Missouri National Guard to implement its distribution plan.
Williams told reporters the plan was the work of a large team of agencies across the state during Thursday’s press conference.
“We have worked diligently for months, even before we received this direction from the CDC, to ensure we are prepared for when the time comes to execute the plans to distribute and administer the vaccine,” Williams said. “We’re grateful to our partners who have shared their knowledge and expertise and will continue to assist us moving forward.”
Williams said the CDC and DHSS began conferring on early planning efforts in April, with multi-agency planning beginning in July. A group of 75 state team members, 50 federal and local partners, and 10 National Guardsman began compiling the 111-page document in August.
Parson said he was proud of the state’s plan and touted it as one other states would look to as a model for their own approaches.
“What this says is that we were prepared before most states in the United States,” Parson said. “We’re going to execute that plan as soon as the vaccine as soon as it gets here. This vaccine will be a huge development moving forward, and we’re going to make sure we can do it as soon as we can and do it right.”
Four vaccines are currently in the third phase of testing, according to Williams. Federal agencies are to announce when a vaccine is approved by the Food and Drug Administration (FDA).
The National Governors Association asked President Trump Thursday for guidance on how states would work with Washington when a vaccine is approved. Specifically, the group sought clarification on expectations for distribution and implementation.
EDITOR’S NOTE: For up-to-date information on coronavirus, check with the CDC and DHSS.

Cameron Gerber studied journalism at Lincoln University. Prior to Lincoln, he earned an associate’s degree from State Fair Community College. Cameron is a native of Eldon, Missouri.
Contact Cameron at cameron@themissouritimes.com.